Copper Plate for Electrochemistry Experiments
৳ 120
- Copper Plate for Chemistry Lab
- Size: 125x50mm
- Made in India

Copper Plate for Electrochemistry Experiments
৳ 120Request a Call Back
- Size Guide
Size Guide
Size Chest Waist Hips XS 34 28 34 S 36 30 36 M 38 32 38 L 40 34 40 XL 42 36 42 2XL 44 38 44 All measurements are in INCHES
and may vary a half inch in either direction.
Size Chest Waist Hips 2XS 32 26 32 XS 34 28 34 S 36 30 36 M 38 32 38 L 40 34 40 XL 42 36 42 All measurements are in INCHES
and may vary a half inch in either direction.
Size Chest Waist Hips XS 34 28 34 S 36 30 36 M 38 32 38 L 40 34 40 XL 42 36 42 2XL 44 38 44 All measurements are in INCHES
and may vary a half inch in either direction.
- Delivery & Return
Delivery
We ship to all 50 states, Washington DC.
All orders are shipped with a UPS tracking number.
Always free shipping for orders over US $200.
During sale periods and promotions the delivery time may be longer than normal.
Return
Elessi will accept exchanges and returns of unworn and unwashed garments within 30 days of the date of purchase (14 days during the sales period), on presentation of the original till receipt at any store where the corresponding collection is available within the country of purchase.
Your return will usually be processed within a week to a week and a half. We’ll send you a Return Notification email to notify you once the return has been completed.
Please allow 1-3 business days for refunds to be received to the original form of payment once the return has been processed.Help
Give us a shout if you have any other questions and/or concerns.
Email: contact@mydomain.com
Phone: +1 (23) 456 789 - Ask a Question

Copper Plate for Electrochemistry Experiments
৳ 120Ask a Question
Description
Electrode Plate Copper with Terminal for chemistry Lab Use. For electrochemistry experiments. 125x50mm plate with socket terminal. This is the best quality Copper Plate for Chemical Experiments mainly used in school and college laboratories in our country.
A copper plate is frequently used in electrochemistry experiments due to copper’s conductive properties, stability, and involvement in redox reactions. Copper is commonly employed as an electrode in various types of electrochemical cells.
Key Uses of a Copper Plate in Electrochemistry:
- Electrode in Galvanic (Voltaic) Cells:
- Copper often serves as the cathode in galvanic cells. The cathode is where reduction occurs, meaning that electrons are gained by the copper ions in the solution (Cu2++2e−→Cutext{Cu}^{2+} + 2e^- rightarrow text{Cu}Cu2++2e−→Cu).
- Daniell Cell Example: Copper is used as the cathode, where copper ions in solution (Cu2+text{Cu}^{2+}Cu2+) are reduced to solid copper.
- Electrodeposition and Electroplating:
- Copper is commonly used in electroplating to deposit a thin layer of copper onto another metal surface. In this process, the copper plate acts as the anode, where it dissolves into the solution as copper ions, which then deposit onto the cathode (the object being plated).
- This process is used for decorative purposes, corrosion protection, or improving electrical conductivity.
- Cathode in Electrolysis:
- In electrolysis, copper plates can act as the cathode where ions from the electrolyte are reduced and deposited. For example, in copper refining, impure copper plates are used as the anode, and pure copper plates as the cathode, allowing pure copper to be deposited on the cathode.
- Corrosion Studies:
- Copper’s behavior in different electrolytes is studied in corrosion science. Since copper is less reactive than metals like zinc, it doesn’t corrode as quickly, making it useful in various electrochemical tests and in constructing corrosion-resistant materials.
- Standard Electrode Potential:
- Copper has a relatively high standard electrode potential (+0.34 V), making it a good reference for determining the relative voltages in electrochemical cells when paired with more reactive metals such as zinc.
Characteristics of Copper Plates in Electrochemical Setups:
- Conductivity: Copper is an excellent conductor of electricity, making it ideal for transferring electrons in an electrochemical circuit.
- Stability: Unlike more reactive metals like zinc, copper is relatively stable and resistant to corrosion, making it a durable choice in electrochemical setups.
- Reduction Reaction: In many galvanic cells, copper serves as the site where electrons are received and copper ions from the solution are reduced.
- Air Protection Product
- Civil Laboratory Equipment
- Aggregate Testing
- Asphalt Testing Equipment
- Bitumen Testing Apparatus
- Cement Testing
- Civil Chemical
- Compression Testing Machine
- Concrete test Apparatus
- Concrete Testing Equipment
- construction products
- Dial Gauge
- Electric Mini Hoist
- Lifting Belt
- Marshall Testing
- Micrometer
- Rebound Hammer
- Sieve Cleaning Brush
- Tension and Compression Testing
- Test Sieve
- Tilting Flume
- Triaxial Machine
- Triple Beam Balance
- Universal Testing Machine
- Distil Water Plant
- Educational Equipment
- Educational Instrument
- Electronics Equipment
- Glass Meter
- Hardware and Tools
- Laboratory Item
- Medical Lab Equipment
- RTC
- RTC Plastic
- Safety Equipment
- Scientific Instrument
- Soil Testing Equipment
- Surveying Equipment
- Test Instruments
- Testing Kit
- সুপারি পাতার পন্য
- Adjustable Pipette, Micropipette
- Beakers, Glass Beaker
- Beakers, Plastic Beaker
- Biggan Bakso, Biggan Box Price in Bangladesh, Educational Lab Apparatus in BD, Science Box Onnorokom
- Biofloc Equipment List, Biofloc Solution, Buffer Solution, Chemical Price in BD, Lab Chemicals
- Biofloc Equipment List, Biofloc Solution, Digital pH Meter, Multi-Purpose Meter, PH Meters, TDS Meters, Water Multi-Parameter, Water Technology & Research
- Biofloc Equipment List, Biofloc Solution, Digital pH Meter, PH Meters, pH Tester, Pocket PH Meter, Water Technology & Research
- BOD Bottle, Glass BOD Bottle, Glass Bottle
- Capacitors, Electronics Lab Equipment in Bangladesh
- Capacity GAZI WASTE BIN
- Centrifuge Machine, Lab Centrifuge, Laboratory Equipment Price in BD
- Conical Flask, Conical Flask in Bangladesh, Glass Conical Flask
- Core, Lab Glassware, Volumetric Flask
- Digital Lux Meter, Light Meter, Lux Meter
- Digital Multimeters, Multimeters
- Dropping Bottle, Glass Bottle, Glass Dropper Bottle
- Educational Lab Apparatus in BD
- Electronics Lab Equipment in Bangladesh, General ICs, IC, Integrated Circuits
- Electronics Lab Equipment in Bangladesh, Transistors
- Electronics Lab Equipment in Bangladesh, Voltage Regulator
- Filter Papers, Whatman Filter Paper
- Gilson Sieves
- Glass Bottle, Lab Bottle, Reagent Bottle
- Glass Bottle, Lab Glassware, Reagent Bottle
- Glass Funnel, Glass Funnel for Lab
- Glass Magnifier, Magnifying Glasses, Professional Magnifier
- Glass Measuring Cylinder, Measuring Cylinder
- Glass Pipette, Lab Glassware
- Laboratory Sieves
- Laboratory Test Sieves
- Lab Sieves
- laser distance measurer
- Measuring Cylinder, Plastic Measuring Cylinder
- measuring tape
- Petri Dish, Plastic Petri Dish
- Pipe Connector, Plastic Connector
- Pipette Pump, Plastic Pump
- Plastic Reagent Bottle, Reagent Bottle
- Refractometer
- Scientific Book Price in Bangladesh
- Sieve Analysis Astm
- Sieves Analysis
- Sieves Test
- Standard Testing Sieve
- Testing Sieve
Related Products
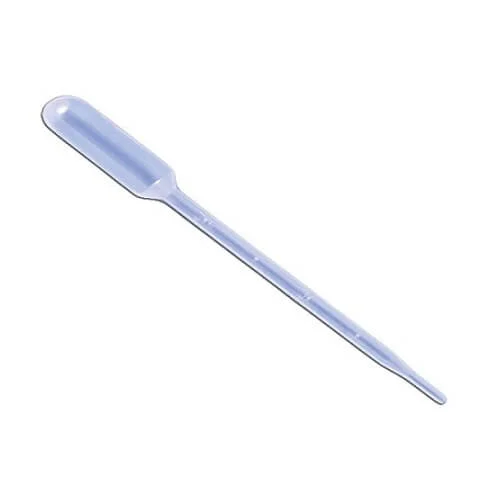
- Laboratory Plastic Dropper
- Transparent Polypropylene
- Marking: 3mL
- Capacity: 5mL
- Made in China
- Digital Photo Tacho and Speed Testing Meter
- Model: DT-2234B
- Made in China
- Digital Temperature and Humidity Hygrometer with Clock
- Model: PH1000
- Brand: Zeal
- Origin: England
- AATCC Crocking Cloth
- Pack Size: 1000 Pieces Box
- Brand: Testfabrics
- Made in the USA.
- Hydrometer, heavy liquid, 0 – 70° ,1000-2000
- Brand: Omsong
- Measuring Range: 0 – 70° ,1000-2000
- Made in : India
- GSM Cutter Blade
- Strong and Durable
- Brand: James Heal
- Made in the UK
- Digital TDS Meter
- TDS Tester with Solution & Auto Calibration
- Model: HI98301 DiST1
- Brand: Hanna
- Made in Romania
- Yellow Fabric Marker or Textile Marker Pen
- Brand: Dalo
- Made in USA.
- এই পণ্যটি আমাদের রেডি স্টকে থাকে না, অর্ডার দিলে ২-৩ দিনের মধ্যে ডেলিভারি করা হবে।
- 4 Digit Analytical Balance
- Model: ATY-224R
- Brand: Shimadzu
- Made in Philipine